Sodium-glucose cotransporter-2 inhibitors (SGLT2i) have been proven to cut back cardiovascular rehospitalisation in coronary heart failure with decreased ejection fraction (HFrEF) sufferers. Nonetheless, it’s unknown whether or not initiating SGLT2i throughout an inpatient keep for a HFrEF exacerbation leads to higher outcomes versus initiation post-discharge in a cohort of diabetic and non-diabetic sufferers. This research compares cardiovascular rehospitalisation, coronary heart failure particular rehospitalisation, cardiovascular dying, and all-cause dying between sufferers initiated on SGLT2i as an inpatient versus post-discharge.
A retrospective research of 4 hospitals in England involving 184 sufferers with HFrEF exacerbations between March 2021 and June 2022 was carried out. Cardiovascular rehospitalisation, coronary heart failure particular rehospitalisation, cardiovascular dying, and all-cause dying had been in contrast between the 2 teams utilizing Cox regression. A Cox proportional-hazards mannequin was fitted to find out predictors of cardiovascular rehospitalisation.
There have been 148 (80.4%) people who acquired SGLT2i as an inpatient, whereas 36 (19.6%) people acquired SGLT2i post-discharge. Median follow-up was 6.5 months for inpatients and seven.5 months for post-discharge sufferers (p=0.522). SGLT2i inpatients had considerably decreased cardiovascular rehospitalisations (22.3%) versus post-discharge sufferers (44.4%) (p=0.005), and considerably decreased coronary heart failure particular rehospitalisations (10.1%) versus post-discharge sufferers (27.8%) (p=0.018). There was no important distinction in all-cause dying (p=0.743) and cardiovascular dying (p=0.816) between the 2 teams. Initiating SGLT2i post-discharge was an unbiased predictor of cardiovascular rehospitalisation (hazard ratio 2.40, 95% confidence interval 1.31 to 4.41, p=0.005).
In conclusion, inpatient SGLT2i initiation for HFrEF exacerbations might cut back cardiovascular and coronary heart failure particular rehospitalisation versus initiation post-discharge. Within the absence of contraindications, clinicians ought to take into account initiating SGLT2i as soon as sufferers are clinically steady throughout inpatient HFrEF admissions.
Introduction
Coronary heart failure with decreased ejection fraction (HFrEF) is a big healthcare burden internationally, with an age-standardised prevalence of roughly 3.8% in ladies and 4.6% in males, and an estimated five-year mortality charge of 43%.1,2 Hospitalisations for coronary heart failure exacerbations symbolize a serious monetary problem to well being companies, and such sufferers have a better threat of readmission and mortality.3
Beforehand, the DAPA-HF (Dapagliflozin in Sufferers with Coronary heart Failure and Diminished Ejection Fraction) trial demonstrated that dapagliflozin considerably reduces the danger of worsening coronary heart failure and cardiovascular dying.4 Equally, the EMPULSE (Empagliflozin in Sufferers Hospitalised for Acute Coronary heart Failure) trial demonstrated that empagliflozin is each protected and offers important scientific profit versus placebo.5 Dapagliflozin is a sodium-glucose cotransporter-2 inhibitor (SGLT2i); the cardioprotective advantages of SGLT2i are postulated to be because of their natriuretic impact, along with blood strain discount, sympathetic nervous system inhibition and improved cardiac vitality metabolism.6–8
The 2021 European Society of Cardiology (ESC) tips advocate that SGLT2i must be commenced previous to discharge from an acute HFrEF exacerbation.9 Nonetheless, it’s at present unknown whether or not inpatient initiation of SGLT2i leads to higher outcomes versus initiation post-discharge in acute HFrEF in a cohort of diabetic and non-diabetic sufferers. Due to this fact, this research goals to match cardiovascular rehospitalisation charges, coronary heart failure particular rehospitalisation charges, cardiovascular dying, and all-cause dying between HFrEF sufferers commenced on SGLT2i as an inpatient versus post-discharge.
Supplies and technique
Inclusion/exclusion standards
This multi-centre retrospective research concerned 184 HFrEF sufferers over 18 years of age, with HFrEF outlined as a left ventricular ejection fraction (LVEF) ≤40%. All sufferers introduced with a New York Coronary heart Affiliation (NYHA) class between II and IV. Sufferers had been admitted with an acute coronary heart failure exacerbation between March 2021 and June 2022. The exclusion standards included sufferers who had been already taking an SGLT2i on admission, identified with sort 1 diabetes mellitus, symptomatic hypotension or had a glomerular filtration charge (GFR) of <20 ml/min/1.73 m2.
Pattern
This research concerned 4 hospitals within the UK. There have been 198 HFrEF sufferers screened, and 14 sufferers had been excluded: 10 had been already taking an SGLT2i on admission, one affected person had symptomatic hypotension, one affected person stopped SGLT2i upon hospital admission, one affected person had a GFR <20 ml/min/1.73 m2 and there have been no knowledge obtainable for one affected person. Due to this fact, 184 sufferers had been included within the last evaluation.
Sufferers had been divided into two teams: sufferers handled with SGLT2i as an inpatient (N=148) and sufferers handled with SGLT2i post-discharge (N=36). An inpatient was outlined as commencing SGLT2i whereas in hospital for an acute HFrEF exacerbation, whereas post-discharge was outlined as commencing SGLT2i after a hospital admission for an acute HFrEF exacerbation. All knowledge had been obtained from the Nationwide Institute for Cardiovascular Outcomes Analysis (NICOR) Coronary heart Failure Audit and digital affected person data, and absolutely anonymised.
Statistical evaluation
The co-primary outcomes of this research had been cardiovascular rehospitalisation and coronary heart failure particular rehospitalisation. Secondary outcomes included cardiovascular dying and all-cause dying. Cardiovascular rehospitalisation was outlined as an unplanned in a single day hospital keep because of a cardiovascular cause.
Baseline demographic, scientific and laboratory parameters had been in contrast between the 2 teams utilizing the Mann-Whitney U take a look at for non-parametric steady variables. The chi-square take a look at was used to match categorical variables between the 2 teams, besides the place greater than 20% of values had been lower than 5 sufferers, wherein case the Fisher’s precise take a look at was used. The normality of steady variables was assessed utilizing the Shapiro-Wilk take a look at. Survival evaluation was carried out utilizing the Kaplan-Meier technique, and the log-rank take a look at was used to match cardiovascular rehospitalisation, all-cause dying and cardiovascular dying between the 2 teams. Cox regression was carried out to match the cardiovascular rehospitalisation charge, coronary heart failure particular rehospitalisation charge, all-cause dying and cardiovascular dying between the 2 teams. Moreover, a Cox proportional-hazards regression mannequin was fitted to find out predictors of cardiovascular rehospitalisation, wherein affected person group, angiotensin-receptor/neprilysin inhibitor (ARNI) utilization, diabetes mellitus, age, intercourse and follow-up time had been covariates. The number of these covariates was based mostly on scientific significance. All statistical analyses had been two-sided, with a significance worth of p<0.05.
All statistical analyses had been carried out on IBM® SPSS® Statistics model 29 (Worldwide Enterprise Machines Company, Armonk, NY, USA).
Affected person and public involvement
Sufferers and the general public weren’t concerned within the design and conduct of this research.
Outcomes
Baseline demographics
Total, 184 sufferers had been included within the research, of which 148 (80.4%) got SGLT2i throughout their inpatient keep, whereas 36 (19.6%) got SGLT2i post-discharge. There have been 182 sufferers prescribed dapagliflozin, whereas two sufferers had been prescribed empagliflozin. Of the SGLT2i post-discharge sufferers, 94.4% (34/36) acquired SGLT2i instantly post-discharge, i.e. the medicine was added to the discharge abstract to be commenced as soon as out of hospital, whereas two sufferers acquired SGLT2i 132 days and 77 days post-discharge, respectively.
The median follow-up of SGLT2i inpatients was 6.5 months and SGLT2i post-discharge sufferers was 7.5 months (p=0.522). All dapagliflozin sufferers had been prescribed 10 mg as soon as every day, besides one affected person who was prescribed 5 mg as soon as every day. The 2 empagliflozin sufferers had been each prescribed 10 mg as soon as every day.
There was no important distinction within the age of SGLT2i inpatients (imply 64.2 ± 14.4 years), and SGLT2i post-discharge sufferers (66.9 ± 12.4 years) (p=0.503). There was additionally no important distinction within the intercourse distribution between SGLT2i inpatients (74.3% male) and SGLT2i post-discharge sufferers (66.7% male) (p=0.354) (desk 1).
Desk 1. Baseline demographics, comorbidities and coverings
SGLT2i inpatient (N=148, 80.4%)
SGLT2i post-discharge affected person(N=36, 19.6%)
p worth
Imply age ± SD, years
64.2 ± 14.4
66.9 ± 12.4
0.503
Male, n (%)
110 (74.3%)
24 (66.7%)
0.354
Median follow-up (IQR), months
6.5 (4.0–11.1)
7.5 (5.0–10.9)
0.522
Ethnicity, n (%)
WhiteMixedAsian/Asian BritishAfrican/CaribbeanOtherUnknown
65 (43.9%)1 (0.7%)32 (21.6%)30 (20.3%)19 (12.8%)1 (0.7%)
16 (44.4%)0 (0.0%)8 (22.2%)6 (16.7%)5 (13.9%)1 (2.8%)
0.848
Comorbidities, n (%)
IHDValve diseaseCongenital coronary heart diseaseHypertensionDMCVACOPDAtrial fibrillation/flutterCardiomyopathy
39 (26.4%)19 (12.8%)1 (0.7%)90 (60.8%)67 (45.3%)12 (8.1%)11 (7.4%)58 (39.2%)39 (26.4%)
11 (30.6%)3 (8.3%)0 (0.0%)28 (77.8%)19 (52.8%)1 (2.8%)3 (8.3%)13 (36.1%)9 (25.0%)
0.6110.4551.0000.0570.4180.2630.7390.7340.868
Smoking Present Ex
11 (7.4%)21 (14.2%)
2 (5.6%)6 (16.7%)
0.875
Malignancy Earlier Present Amyloid
5 (3.4%)9 (6.1%)1 (0.7%)
1 (2.8%)2 (5.6%)0 (0.0%)
0.976 1.000
Gadget remedy, n (%)
0.253
CRT-DCRT-PICDPM
9 (6.1%)2 (1.4%)17 (11.5%)8 (5.4%)
5 (13.9%)1 (2.8%)1 (2.8%)1 (2.8%)
Drugs, n (%)
ACEiARBBeta blockerLoop diureticThiazide diureticMRAAspirinOther antiplateletDigoxinStatinWarfarinOther oral anticoagulantAmiodaroneISMNIvabradineARNI
76 (51.4%)24 (16.2%)141 (95.3%)132 (89.2%)4 (2.7%)117 (79.1%)21 (14.2%)16 (10.8%)27 (18.2%)60 (40.5%)14 (9.5%)65 (43.9%)5 (3.4%)7 (4.7%)16 (10.8%)37 (25.0%)
20 (55.6%)7 (19.4%)32 (88.9%)30 (83.3%)1 (2.8%)26 (72.2%)8 (22.2%)2 (5.6%)4 (11.1%)19 (52.8%)3 (8.3%)11 (30.6%)2 (5.6%)1 (2.8%)2 (5.6%)5 (13.9%)
0.6510.6430.1480.3311.0000.3770.2350.3410.3050.1830.8340.1440.6240.6070.3410.154
Chi-squared, Fisher’s precise, and Mann-Whitney U checks had been used to match variables between SGLT2i inpatients versus post-discharge sufferers.Key: ACEi = angiotensin-converting enzyme inhibitor; ARB = angiotensin-receptor blocker; ARNI = angiotensin-receptor/neprilysin inhibitor; CCB = calcium-channel blocker; COPD = continual obstructive pulmonary illness; CRT-D = cardiac resynchronisation therapy-defibrillator; CRT-P = cardiac resynchronisation therapy-pacemaker; CVA = cerebrovascular accident; DM = diabetes mellitus; ICD = implantable cardioverter defibrillator; IHD = ischaemic coronary heart illness; IQR = interquartile vary; ISMN = isosorbide mononitrate; MRA = mineralocorticoid receptor antagonist; PM = pacemaker; SD = normal deviation; SGLT2i = sodium-glucose cotransporter-2 inhibitor
The distribution of ethnicities and comorbidities had been related between the 2 teams (all p>0.05). There was additionally no important distinction within the proportion of sufferers on system therapies and all related cardiac drugs (all p>0.05) (desk 1).
The imply LVEF of SGLT2i inpatients (26.7%) and SGLT2i post-discharge sufferers (26.2%) had been related (p=0.749). Moreover, there was no important distinction within the distribution of NYHA lessons between SGLT2i inpatients and SGLT2i post-discharge sufferers (p=0.591). Laboratory parameters weren’t considerably totally different between SGLT2i inpatients and SGLT2i post-discharge sufferers (p>0.05), aside from complete ldl cholesterol, wherein SGLT2i post-discharge sufferers had considerably higher values (p=0.030) (desk 2).
Desk 2. Baseline scientific traits and laboratory values of research sufferers
SGLT2i inpatient (N=148, 80.4%)
SGLT2i post-discharge affected person(N=36, 19.6%)
p worth
Scientific traits, imply (vary)
Coronary heart charge, bpmSystolic BP, mmHgLVEF, % <10, n (%) 10–19, n (%) 20–29, n (%) 30–40, n (%) Unknown, n (%)NYHA, n (%)Class IIClass IIIClass IVUnknown
75.7 (50–127)114.2 (77–176)26.7 (9–40) 1 (0.7%) 24 (16.2%) 58 (39.2%) 64 (43.2%) 1 (0.7%) 76 (51.4%)25 (16.9%)7 (4.7%)40 (27.0%)
78.9 (59–114)117.7 (97–148)26.2 (10–40) 0 (0.0%) 5 (13.9%) 15 (41.7%) 15 (41.7%) 1 (2.8%) 23 (63.9%)5 (13.9%)0 (0.0%)8 (22.2%)
0.2460.0850.653 0.591
Laboratory parameter, imply (vary)
Glucose, mmol/lHbA1c, mmol/molTotal ldl cholesterol, mmol/lLDL-cholesterol, mmol/lHDL-cholesterol, mmol/lTriglyceride, mmol/lUrea, mmol/lCreatinine, mmol/l
8.3 (3.8–25.7)50.1 (18–144)3.8 (1.5–8.5)2.0 (0.6–5.6)1.1 (0.3–2.5)1.6 (0.4–7.6)9.2 (3.2–25.2)113.9 (55–207)
8.0 (4.1–21.7)56.9 (33–139)4.3 (2.6–6.4)2.2 (0.9–4.1)1.3 (0.7–2.4)1.9 (0.4–5.3)8.3 (3.7–19.9)108.7 (50–214)
0.7260.3520.0300.1740.1030.0550.1870.327
Chi-squared, Fisher’s precise, and Mann-Whitney U checks had been used to match variables between SGLT2i inpatients versus post-discharge sufferers. Vital values are highlighted in daring.Key: BP = blood strain; bpm = beats per minute; HbA1c = glycated haemoglobin; HDL = high-density lipoprotein; LDL = low-density lipoprotein; NYHA = New York Coronary heart Affiliation; SGLT2i = sodium-glucose cotransporter-2 inhibitor
Survival evaluation
Total, the cardiovascular rehospitalisation charge was considerably decrease in SGLT2i inpatients (33/148, 22.3%) versus SGLT2i post-discharge sufferers (16/36, 44.4%) (hazard ratio [HR] 2.30, 95% confidence interval [CI] 1.27 to 4.19, p=0.006). Moreover, the center failure particular rehospitalisation charge was considerably decrease in SGLT2i inpatients (15/148, 10.1%) versus SGLT2i post-discharge sufferers (10/36, 27.8%) (HR 2.71, 95percentCI 1.19 to six.19, p=0.018). Nonetheless, there have been no important variations in cardiovascular dying (HR 1.20, 95percentCI 0.25 to five.80, p=0.817) and all-cause dying (HR 1.20, 95percentCI 0.40 to three.66, p=0.744) between the 2 teams (desk 3).
Desk 3. Scientific finish factors
Scientific finish level, n (%)
SGLT2i inpatient(N=148, 80.4%)
SGLT2i post-discharge affected person(N=36, 19.6%)
Hazard ratio (95percentCI)
p worth
Cardiovascular rehospitalisations
33 (22.3%)
16 (44.4%)
2.30 (1.27 to 4.19)
0.006
Coronary heart failure particular rehospitalisations
15 (10.1%)
10 (27.8%)
2.71 (1.19 to six.19)
0.018
Cardiovascular dying
8 (5.4%)
2 (5.6%)
1.20 (0.25 to five.80)
0.817
All-cause dying
14 (9.5%)
4 (11.1%)
1.20 (0.40 to three.66)
0.744
Cox regression was used to match outcomes between SGLT2i inpatients versus post-discharge sufferers. Vital values are highlighted in daring.Key: CI = confidence interval; SGLT2i = sodium-glucose cotransporter-2 inhibitor
The log-rank take a look at additionally demonstrated that cardiovascular rehospitalisation was considerably decreased in SGLT2i inpatients versus SGLT2i post-discharge sufferers (log-rank p=0.005, determine 1). Within the SGLT2i inpatient group, causes of cardiovascular rehospitalisation included: 45.5% (15/33) from coronary heart failure exacerbation, 9.1% (3/33) from cardiogenic shock, 9.1% (3/33) from stroke, 6.1% (2/33) from cardiogenic syncope, 6.1% (2/33) from non-ST elevation myocardial infarction, 6.1% (2/33) from cardiac arrest, 3.0% (1/33) from ventricular bigeminy, 3.0% (1/33) from palpitations, 3.0% (1/33) from atrial fibrillation, 3.0% (1/33) from aortic stenosis, and three.0% (1/33) from sepsis. The reason for rehospitalisation of 1 affected person on this group was unknown. Within the SGLT2i post-discharge sufferers, causes of cardiovascular rehospitalisation included: 62.5% (10/16) from coronary heart failure exacerbation, 6.25% (1/16) from anaemia, 6.25% (1/16) from ischaemic coronary heart illness, 6.25% (1/16) from multi-vessel coronary illness, 6.25% (1/16) from aortic stenosis, 6.25% (1/16) from unstable angina and 6.25% (1/16) from stomach aortic aneurysm.
Log-rank statistical testing was used to match the proportion of sufferers avoiding cardiovascular rehospitalisation between inpatients and post-discharge sufferers (p<0.05).
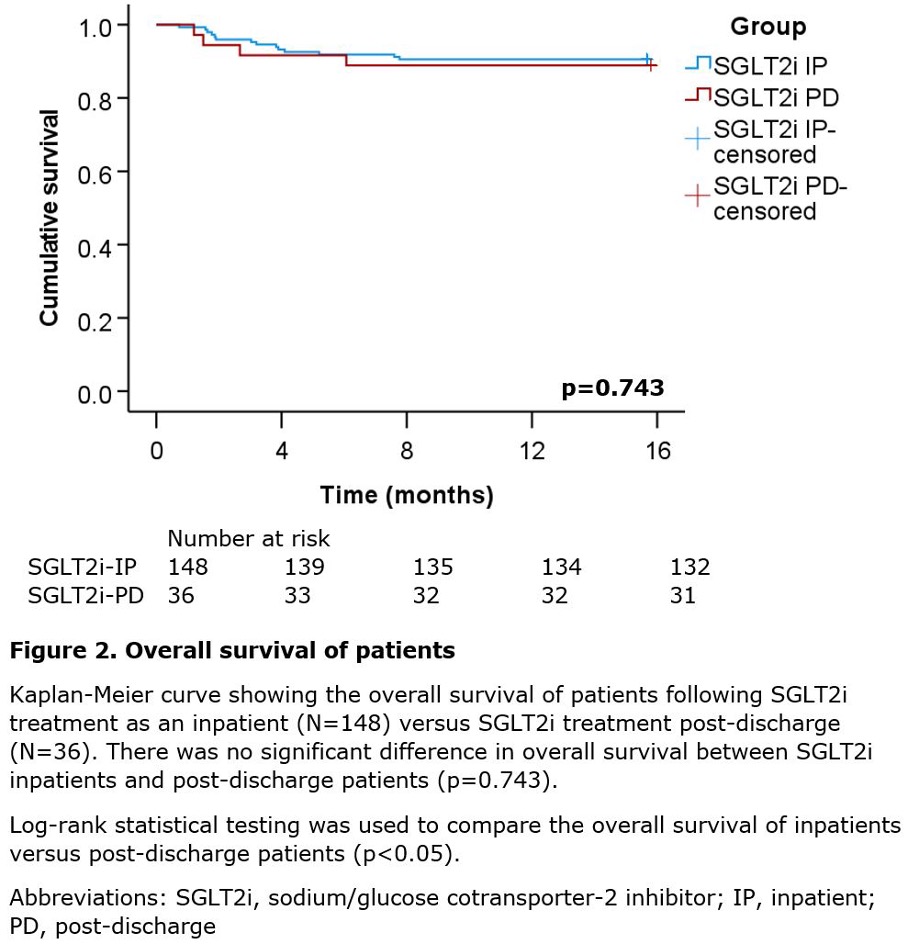
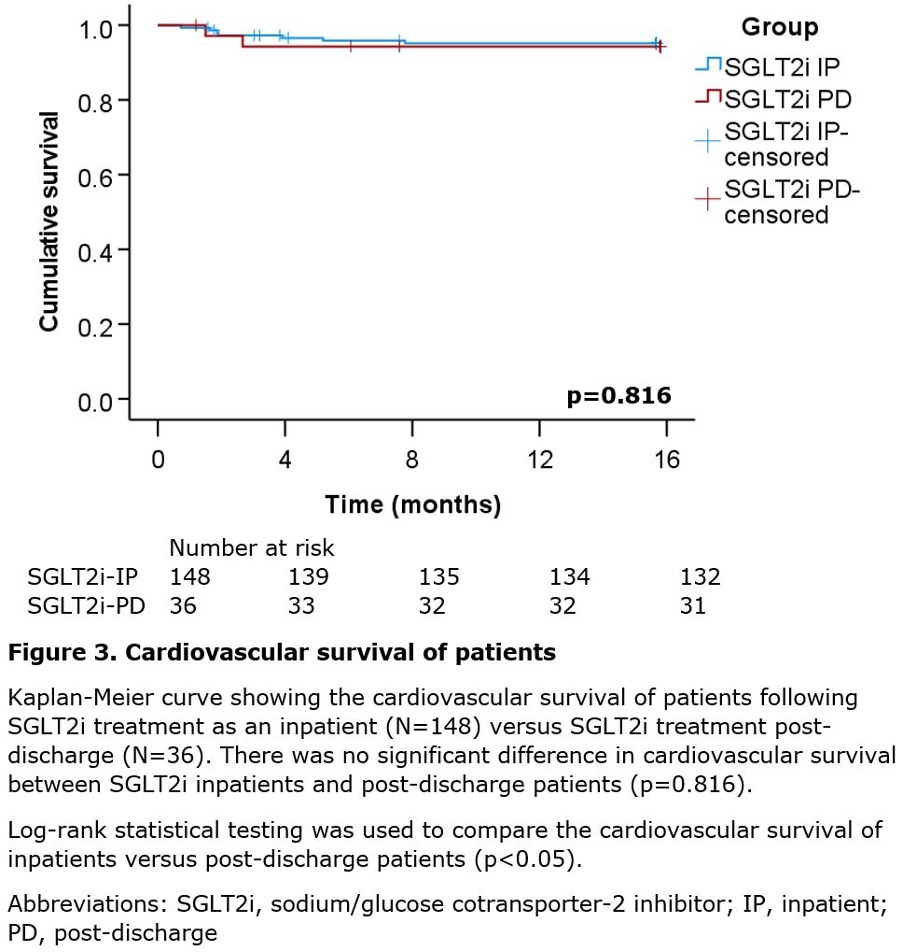
There was no important distinction in all-cause dying (log-rank p=0.743, determine 2) and cardiovascular dying (log-rank p=0.816, determine 3) between SGLT2i inpatients versus SGLT2i post-discharge sufferers. Total, 55.6% (10/18) of deaths had been cardiac associated: seven from coronary heart failure exacerbation, one from pulmonary oedema, one from multi-organ failure secondary to coronary heart failure, and one from cardiac arrest. An extra 11.1% (2/18) of deaths had been non-cardiac and included one case of sepsis and community-acquired pneumonia, and one other case of biliary sepsis. The reason for dying for 33.3% (6/18) of sufferers was unknown.
A Cox proportional-hazards regression mannequin was fitted to find out predictors of cardiovascular rehospitalisation. Receiving SGLT2i post-discharge was an unbiased predictor of cardiovascular rehospitalisation; these sufferers had a considerably elevated threat of cardiovascular rehospitalisation (HR 2.40, 95percentCI 1.31 to 4.41, p=0.005) versus SGLT2i inpatients. ARNI utilization, diabetes mellitus, age, intercourse and follow-up time weren’t important predictors of cardiovascular rehospitalisation (all p>0.05) (desk 4).
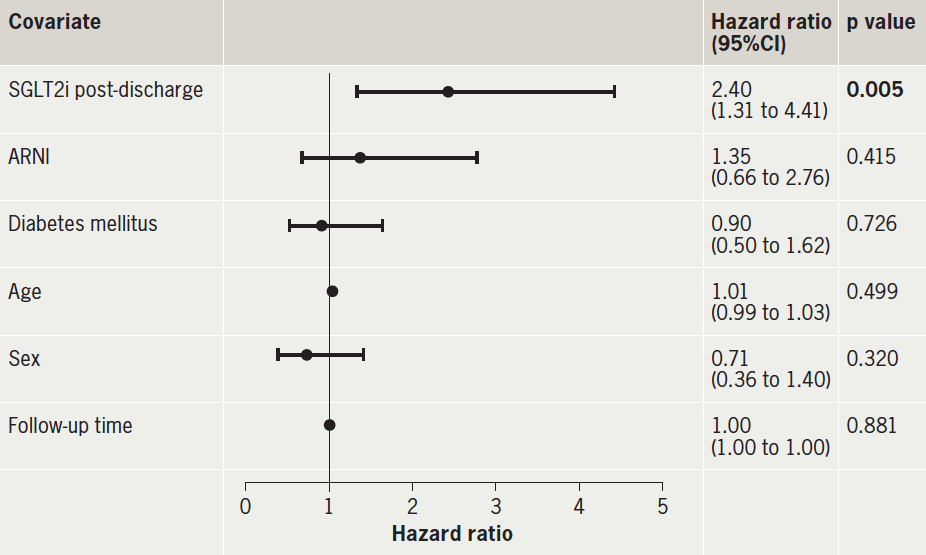
Vital values are highlighted in daring.Key: ARNI = angiotensin-receptor/neprilysin inhibitor; CI = confidence interval; SGLT2i = sodium-glucose cotransporter-2 inhibitor
Dialogue
Beforehand, the STRONG-HF (Security, Tolerability, and Efficacy of Speedy Optimization, Helped by NT-proBNP Testing, of Coronary heart Failure Therapies) trial has demonstrated that fast up-titration of guideline-directed medical remedy considerably reduces coronary heart failure readmission and all-cause dying in HFrEF sufferers versus traditional care. Nonetheless, the STRONG-HF trial was performed previous to the appearance of SGLT2i.10 Moreover, the DAPA-HF trial has demonstrated that dapagliflozin considerably reduces cardiovascular rehospitalisation and cardiovascular dying in HFrEF sufferers.4 Nonetheless, it’s at present unknown whether or not initiating SGLT2i throughout an inpatient keep for an acute HFrEF exacerbation leads to higher outcomes versus initiating SGLT2i post-discharge in a cohort of each diabetic and non-diabetic sufferers. To the authors’ data, that is the primary research to match cardiovascular rehospitalisation, coronary heart failure particular rehospitalisation, cardiovascular dying, and all-cause dying between HFrEF sufferers handled with SGLT2i as an inpatient versus post-discharge in a cohort of each diabetic and non-diabetic sufferers.
The salient findings of this research are as follows. First, initiating SGLT2i throughout an inpatient keep for an acute HFrEF exacerbation considerably lowers cardiovascular rehospitalisation charges versus initiating SGLT2i post-discharge (p=0.006, desk 3). Second, initiating SGLT2i throughout an inpatient keep for an acute HFrEF exacerbation considerably lowers coronary heart failure particular rehospitalisation charges versus initiating SGLT2i post-discharge (p=0.018, desk 3). Third, there isn’t a important distinction in all-cause dying between SGLT2i inpatients and post-discharge sufferers (p=0.743, determine 2). Fourth, there isn’t a important distinction in cardiovascular dying between SGLT2i inpatients and post-discharge sufferers (p=0.816, determine 3).
Beforehand, the Sotagliflozin in Sufferers with Diabetes and Current Worsening Coronary heart Failure (SOLOIST-WHF) trial demonstrated that no matter inpatient or post-discharge initiation of sotagliflozin, there was a big discount within the composite major end result of cardiovascular dying, cardiovascular rehospitalisation and pressing visits because of HFrEF, versus placebo. The authors concluded that SGLT2i must be initiated in sort 2 diabetic sufferers with worsening coronary heart failure as quickly because the affected person is clinically steady, and ideally previous to discharge.11 This research builds on the SOLOIST-WHF trial by demonstrating that this profit extends to non-diabetic sufferers, and in addition displaying {that a} additional profit is achieved by beginning SGLT2i as an inpatient versus post-discharge. Moreover, this research offers assist to the 2021 ESC tips, which advocate that evidence-based oral medical remedy, together with SGLT2i, must be initiated earlier than discharge.9
A possible rationalization for the decreased cardiovascular and coronary heart failure particular rehospitalisation charges in SGLT2i inpatients versus post-discharge sufferers could also be because of synergism between SGLT2i and furosemide. Each medicine act throughout the nephron: whereas furosemide inhibits sodium and, therefore, water reabsorption within the ascending loop of Henle via binding to the sodium-potassium-chloride cotransporter, SGLT2i inhibits SGLT2 transporters within the proximal convoluted tubule to stop sodium, glucose and, subsequently, water reabsorption.8,12 Moreover, Ibrahim et al., reported considerably higher urine output and diuretic response, with no important opposed influence on renal perform, in sufferers managed with SGLT2i and furosemide mixture remedy versus furosemide monotherapy (p<0.001).13 Due to this fact, administering SGLT2i and furosemide together throughout an inpatient keep when the affected person is most acutely fluid overloaded might end in higher outcomes and, therefore, cut back rehospitalisation threat versus remedy post-discharge. Lowering rehospitalisation charges because of HFrEF is significant, as a result of it could cut back the monetary burden on the healthcare system and enhance affected person prognosis.14,15
The outcomes of this research must be interpreted within the context of some limitations. It was a retrospective research, with a comparatively small pattern dimension. Since solely 4 hospitals had been studied, this probably limits the research’s generalisability. Future research ought to think about using the broader NICOR Coronary heart Failure Audit database to extend generalisability. There was a discrepancy within the dimension of the inpatient and the post-discharge teams. Moreover, the size of follow-up might have been too quick to find out the true distinction in all-cause dying and cardiovascular dying between the 2 teams. Nonetheless, this was unavoidable, on condition that the Nationwide Institute for Well being and Care Excellence (NICE) solely really helpful dapagliflozin for HFrEF since late February 2021, and empagliflozin was solely really helpful for HFrEF since March 2022.16,17 Additionally it is vital to acknowledge the inherent limitations of utilising registry knowledge, such because the potential variability in high quality of collected knowledge, and that the researchers didn’t gather the unique knowledge themselves. Future research ought to, thus, incorporate an extended follow-up to find out whether or not all-cause dying and cardiovascular dying are really totally different between SGLT2i inpatients and post-discharge sufferers. A randomised-controlled potential research would additionally construct on this research’s findings and enhance the extent of proof.
The latest PRESERVED-HF (Results of Dapagliflozin on Biomarkers, Signs and Practical Standing in Sufferers With Preserved Ejection Fraction Coronary heart Failure) trial has additionally recommended that SGLT2i might enhance signs and bodily limitations amongst coronary heart failure with preserved ejection fraction (HFpEF) sufferers.18 Due to this fact, it will be fascinating to match outcomes between SGLT2i inpatients and post-discharge sufferers with HFpEF similarly to this research.
In conclusion, this research has demonstrated that initiating SGLT2i throughout an inpatient keep for an acute exacerbation of HFrEF might considerably decrease cardiovascular and coronary heart failure particular rehospitalisation charges versus initiating SGLT2i post-discharge. Due to this fact, within the absence of a contraindication, clinicians ought to take into account initiating SGLT2i as soon as the affected person is clinically steady throughout an inpatient admission of HFrEF, no matter whether or not the affected person has sort 2 diabetes.
Key messages
Earlier research have proven that sodium-glucose cotransporter-2 inhibitors (SGLT2i) cut back cardiovascular rehospitalisation and dying in coronary heart failure with decreased ejection fraction (HFrEF) sufferers
Nonetheless, it’s unknown whether or not initiating a SGLT2i as an inpatient for a HFrEF exacerbation improves outcomes versus initiation post-discharge in a cohort of diabetic and non-diabetic sufferers
This research demonstrated that initiating a SGLT2i for inpatients with a HFrEF exacerbation resulted in considerably decrease cardiovascular and coronary heart failure particular rehospitalisation charges versus initiation post-discharge
Within the absence of contraindications, clinicians ought to take into account initiating a SGLT2i as quickly because the affected person is clinically steady throughout an inpatient admission for HFrEF, no matter whether or not the affected person has sort 2 diabetes
Conflicts of curiosity
None declared.
Funding
None.
Research approval
This research was a retrospective evaluation of registry knowledge for the needs of high quality assurance, subsequently the necessity for formal moral approval was waived by our establishment (Queen Mary, College of London). All knowledge had been absolutely anonymised.
Acknowledgements
At the start, we wish to thank Dr Shanti Velmurugan for overseeing this mission. We might additionally wish to thank all the center failure employees throughout all disciplines in Barts Well being NHS Belief. Lastly, we wish to prolong our gratitude to Mr Patrick Moran from the Barts Well being NHS Belief coronary heart failure audit workforce.
Editors’ notice
Figures 2 and three can be found within the on-line model of this text.
References
1. Tiller D, Russ M, Greiser KH et al. Prevalence of symptomatic coronary heart failure with decreased and with regular ejection fraction in an aged common inhabitants – the CARLA research. PLoS One 2013;8:e59225. https://doi.org/10.1371/journal.pone.0059225
2. Groenewegen A, Rutten FH, Mosterd A, Hoes AW. Epidemiology of coronary heart failure. Eur J Coronary heart Fail 2020;22:1342–56. https://doi.org/10.1002/ejhf.1858
3. Agarwal MA, Fonarow GC, Ziaeian B. Nationwide traits in coronary heart failure hospitalizations and readmissions from 2010 to 2017. JAMA Cardiol 2021;6:952. https://doi.org/10.1001/jamacardio.2020.7472
4. McMurray JJV, Solomon SD, Inzucchi SE et al. Dapagliflozin in sufferers with coronary heart failure and decreased ejection fraction. N Engl J Med 2019;381:1995–2008. https://doi.org/10.1056/NEJMoa1911303
5. Voors AA, Angermann CE, Teerlink JR et al. The SGLT2 inhibitor empagliflozin in sufferers hospitalized for acute coronary heart failure: a multinational randomized trial. Nat Med 2022;28:568–74. https://doi.org/10.1038/s41591-021-01659-1
6. Packer M, Anker SD, Butler J, Filippatos G, Zannad F. Results of sodium-glucose cotransporter 2 inhibitors for the remedy of sufferers with coronary heart failure: proposal of a novel mechanism of motion. JAMA Cardiol 2017;2:1025. https://doi.org/10.1001/jamacardio.2017.2275
7. Lytvyn Y, Bjornstad P, Udell JA, Lovshin JA, Cherney DZI. Sodium glucose cotransporter-2 inhibition in coronary heart failure: potential mechanisms, scientific functions, and abstract of scientific trials. Circulation 2017;136:1643–58. https://doi.org/10.1161/CIRCULATIONAHA.117.030012
8. Lopaschuk GD, Verma S. Mechanisms of cardiovascular advantages of sodium glucose co-transporter 2 (SGLT2) inhibitors. JACC Primary Transl Sci 2020;5:632–44. https://doi.org/10.1016/j.jacbts.2020.02.004
9. McDonagh TA, Metra M, Adamo M et al. 2021 ESC tips for the prognosis and remedy of acute and continual coronary heart failure. Eur Coronary heart J 2021;42:3599–726. https://doi.org/10.1093/eurheartj/ehab368
10. Mebazaa A, Davison B, Chioncel O et al. Security, tolerability and efficacy of up-titration of guideline-directed medical therapies for acute coronary heart failure (STRONG-HF): a multinational, open-label, randomised, trial. Lancet 2022;400:1938–52. https://doi.org/10.1016/S0140-6736(22)02076-1
11. Bhatt DL, Szarek M, Steg PG et al. Sotagliflozin in sufferers with diabetes and up to date worsening coronary heart failure. N Engl J Med 2021;384:117–28. https://doi.org/10.1056/NEJMoa2030183
12. Huxel C, Raja A, Ollivierre-Lawrence MD. Loop diuretics. In: StatPearls. Treasure Island, FL: StatPearls Publishing, 2022. Obtainable from: http://www.ncbi.nlm.nih.gov/books/NBK546656/ [accessed 28 July 2022].
13. Ibrahim A, Ghaleb R, Mansour H et al. Security and efficacy of including dapagliflozin to furosemide in sort 2 diabetic sufferers with decompensated coronary heart failure and decreased ejection fraction. Entrance Cardiovasc Med 2020;7:602251. https://doi.org/10.3389/fcvm.2020.602251
14. Kwok CS, Abramov D, Parwani P et al. Price of inpatient coronary heart failure care and 30-day readmissions in the USA. Int J Cardiol 2021;329:115–22. https://doi.org/10.1016/j.ijcard.2020.12.020
15. Shahar E, Lee S, Kim J, Duval S, Barber C, Luepker RV. Hospitalized coronary heart failure: charges and long-term mortality. J Cardiac Fail 2004;10:374–9. https://doi.org/10.1016/j.cardfail.2004.02.003
16. Nationwide Institute for Well being and Care Excellence. Dapagliflozin for treating continual coronary heart failure with decreased ejection fraction. London: NICE, 2021. Obtainable from: https://www.good.org.uk/steering/ta679
17. Nationwide Institute of Well being and Care Excellence. Empagliflozin for treating continual coronary heart failure with decreased ejection fraction – suggestions. London: NICE, 2022. Obtainable from: https://www.good.org.uk/steering/ta679/chapter/1-Suggestions
18. Nassif ME, Windsor SL, Borlaug BA et al. The SGLT2 inhibitor dapagliflozin in coronary heart failure with preserved ejection fraction: a multicenter randomized trial. Nat Med 2021;27:1954–60. https://doi.org/10.1038/s41591-021-01536-x